You should know from Atomic Structure and Fundamental Particles that atoms are made up of protons, neutrons, and electrons. We mentioned above that bonding occurs because of attraction between positive and negative charges. If these atoms come from two or more different elements, then we call the molecule a compound. They are already as stable as they can be! Instead, we find them as monatomic gases.Ī molecule is made of two or more atoms chemically bonded together. This is why noble gases don’t readily form bonds with other atoms.
ION BONDING FACTS FULL
To be at its most stable, an atom wants to have a full outer shell of electrons, much like that of a noble gas.
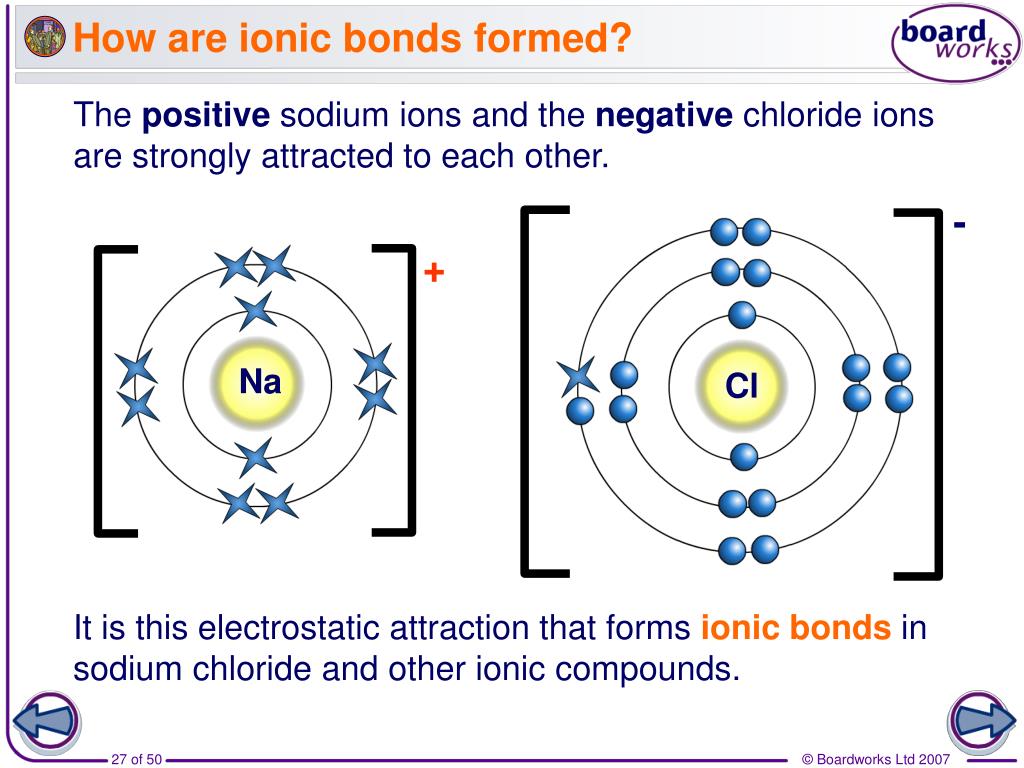
Stability depends on how many electrons an atom has in its outer shell. By combining with other atoms in various combinations, they can form different substances with much lower energy states. Why do bonds form?Ītoms want to be stable - they like being in the lowest energy state possible. When we mention bonding here, we mean primary bonding, unless otherwise stated. They are more commonly known as I ntermolecular Forces, since they take place between molecules. In contrast, secondary bonds are a lot weaker.

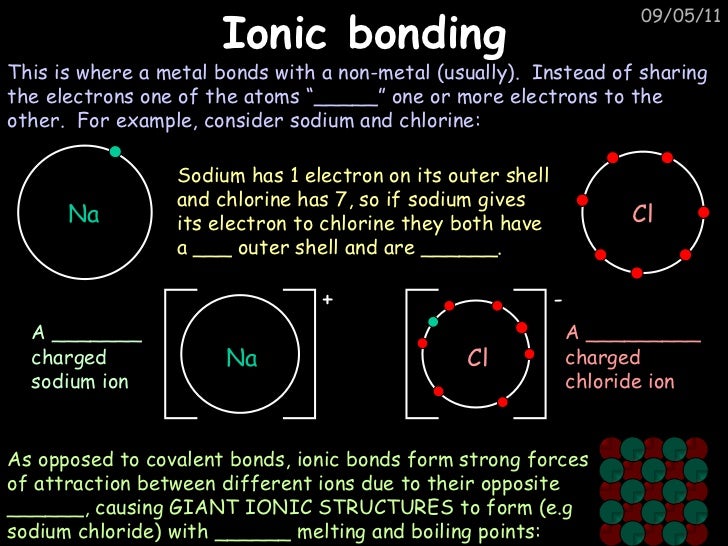
They’re generally strong and hard to break. They take place between atoms within the molecule. Primary bonds are also known as intramolecular forces. Primary bonding is what most people think of when the word is mentioned. There are two categories of bonding, known as primary and secondary bonding. It is caused by the lasting attraction between positive and negative charges. Reaction Quotient and Le Chatelier's Principleīonding is the interaction of different atoms to form compounds, molecules, ions, crystals, and all the other substances that make up the world.
Elemental Composition of Pure Substances.Application of Le Chatelier's Principle.Intramolecular Force and Potential Energy.Variable Oxidation State of Transition Elements.Transition Metal Ions in Aqueous Solution.
